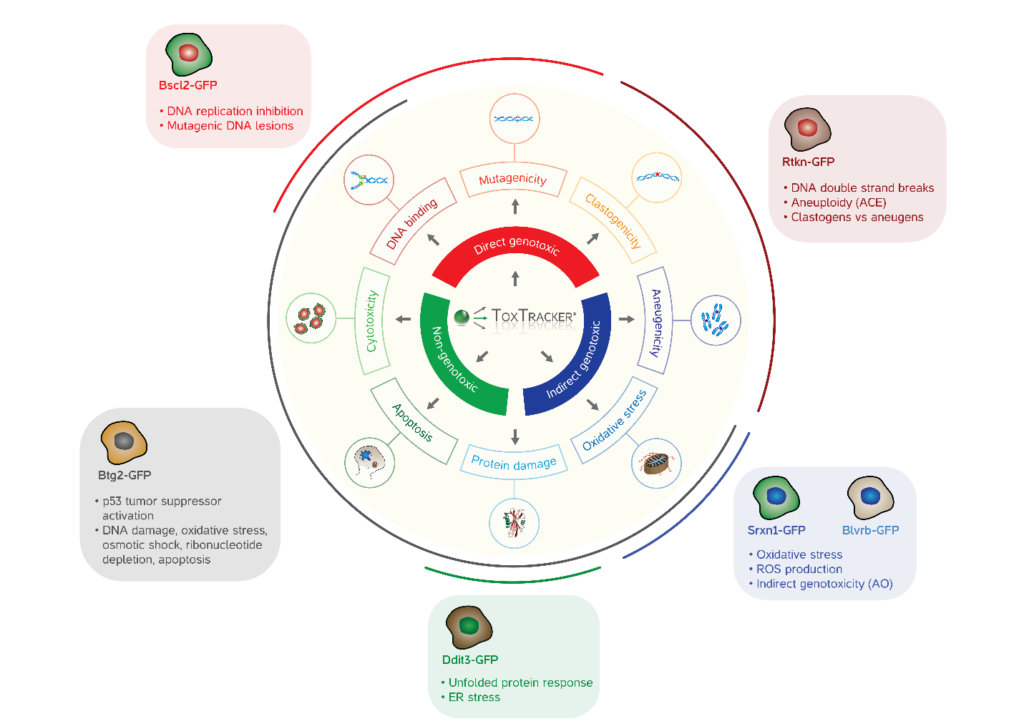
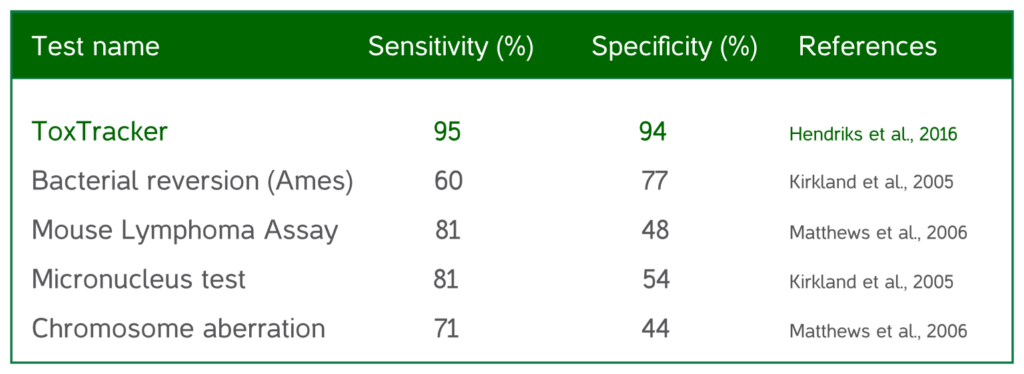
ToxTracker® is a unique, stem-cell based assay which takes advantage of a Green Fluorescent Protein (GFP) reporter system to identify hazard and mode-of-action (MOA) of potentially genotoxic materials. ToxTracker has been extensively validated to predict in vivo genotoxic substances with an accuracy of more than 90%. Over 1,500 compounds have been successfully evaluated with the assay. Parent compounds as well as their metabolites are evaluated for genotoxicity with the inclusion induced-S9 mix in the test system prior to test material exposure.
To identify aneugens and clastogens, the standard ToxTracker assay can be augmented with the ACE protocol, which is able to detect perturbations in cell cycle continuity and polyploidy. Similarly, indirect genotoxicity and direct DNA reactivity MOAs can be resolved with the AO (Antioxidant) extension.
In addition to accurately identifying genotoxins and genotoxicants, ToxTracker will also identify non-genotoxic mechanisms that are associated with carcinogenesis; substances may denature proteins or induce oxidative stress which can lead to neoplasm formation without causing direct DNA damage. Confirmation of this MOA, although not conclusively indicating carcinogenic potential, may be used to select substances for further analysis.
ToxTracker is currently being reviewed by OECD for Test Guideline establishment and results can be submitted to regulatory authorities having oversight of pharmaceuticals under a Weight-of Evidence approach in accordance with ICH S2(R1) and M7 guidelines.
Contact IIVS at clientservices@iivs.org for further information and pricing.